
Partial Reduction of Alkynes With Lindlar’s Catalyst
Partial Reduction of Alkynes to cis Alkenes with the Lindlar Catalyst
- Alkynes are generally more reactive towards catalytic hydrogenation (e.g. Pd-C, H2) than alkenes
- While it’s possible to partially hydrogenate an alkyne to an alkene by limiting the number of equivalents of hydrogen gas, it’s not very convenient (since it requires equpiment such as a gas buret, and is impractical to do on small scales).
- An alternative approach is to make the Pd-catalyst less active for hydrogenation by “poisoning” it, often with lead salts (Pb) and amines (such as quinoline). The combination of Pd supported on calcium carbonate (CaCO3) that has been treated with a small amount of lead (often Pb(OAc)2 ) is known as “Lindlar’s catalyst”.
- Lindlar’s catalyst always gives cis alkenes from alkynes. Alkenes are not hydrogenated.
- Alternative reagents that are equivalent to Lindlar’s catalyst include Pd on barium sulfate (Pd/BaSO4) with quinoline, as well as nickel boride (Ni2B).
- The stereochenistry of this reaction is very commonly tested in exams, expecially in combination with reactions of alkenes that are also stereospecific (e.g. halogenation, dihydroxylation).
- Note that it’s also possible to partially reduce alkynes to trans-alkenes through the use of sodium in ammonia (Na/NH3) - See article - Partial Reduction of Alkynes to Trans Alkenes)
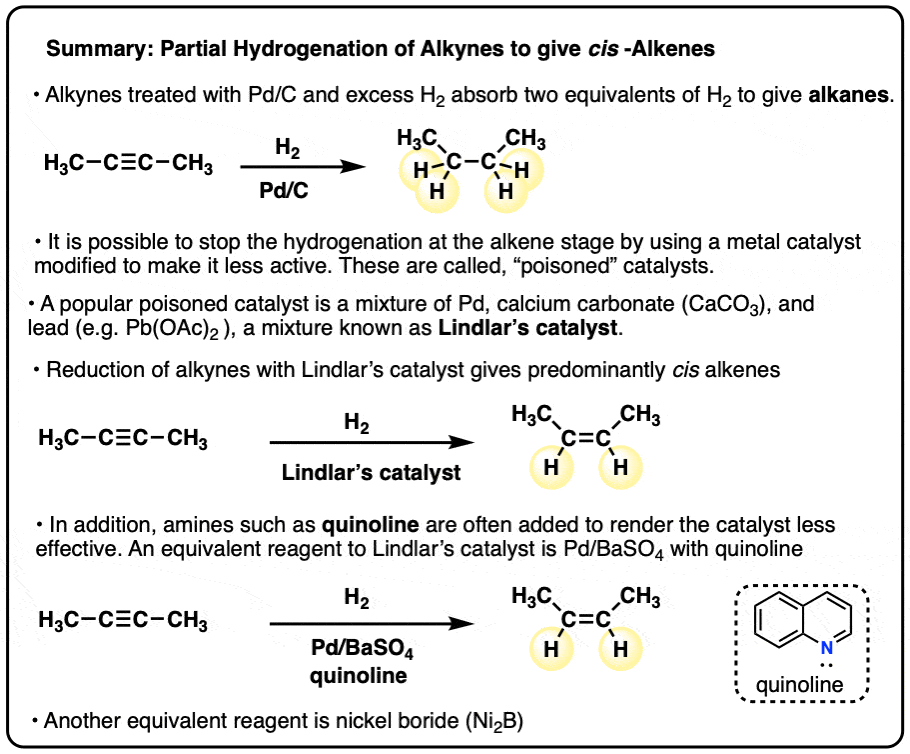
Table of Contents
-
- Partial Hydrogenation of Alkynes to Alkenes With Lindlar’s Catalyst
- Examples of Partial Reduction
- Applications of Lindlar’s Catalyst
- Using Lindlar’s Catalyst in Synthesis
- Summary
- Notes
- Quiz Yourself!
- (Advanced) References and Further Reading
1. Partial Hydrogenation of Alkynes to Alkenes With Lindlar’s Catalyst
We’ve seen that catalytic hydrogenation is a useful reaction for converting alkenes into alkanes. [See article: Catalytic Hydrogenation of Alkenes]
In catalytic hydrogenation, hydrogen gas is adsorbed onto the surface of a “late” metal (usually palladium or platinum) that has been dispersed on a high surface-area material such as activated carbon.
Alkenes and other unsaturated molecules readily bind to late metals through their pi-bonds. When an alkene lands on a metal packed with hydrogen on its surface, an addition reaction occurs where two new C-H bonds are formed and a C-C pi bond is broken, generally with syn stereoselectivity. This produces an alkane.
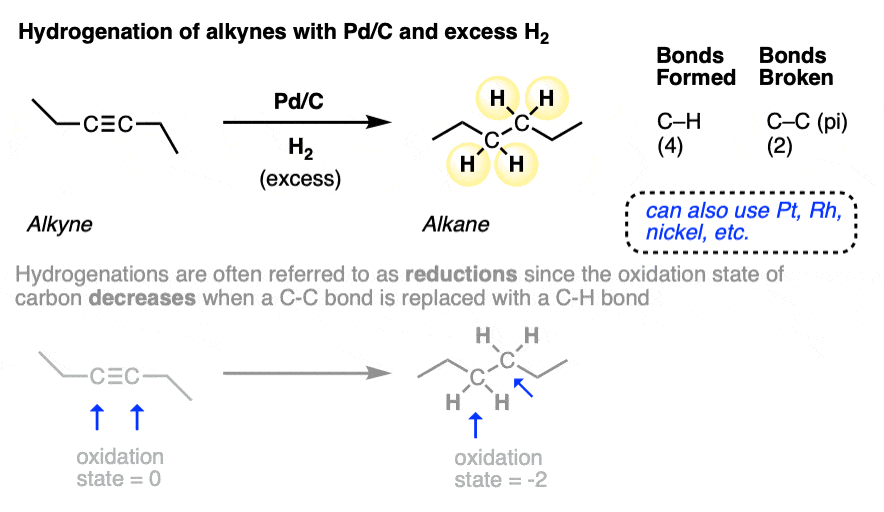
When alkynes are used in place of alkenes in the presence of Pd-C and H2, both double bonds are hydrogenated, also resulting in an alkane:
This reaction is often referred to as a “reduction” since the oxidation state of carbon is lowered during this process (oxidation state of each carbon in the neutral alkyne is zero; oxidation state of each carbon in the alkane is -2. See article - Calculating the Oxidation State of Carbon).
Forgive me for my lack of excitement, but I think the proper reaction to this particular reaction is, “big whoop”.
I mean, making alkanes from alkanes is fine if your plan is to make margarine or something. But from a chemical synthesis perspective, it’s a waste of a potentially useful functional group.
Why? Well, we’ve just seen that acetylide ions are extremely useful for making C-C bonds through reaction (SN2) with alkyl halides. (See article - Substitution Reactions of Acetylides). For the first time, this gave us a method for building up longer carbon chains from smaller ones.
Using this reaction just for making longer alkanes isn’t exactly exciting because there aren’t that many interesting reactions of alkanes aside from 1) free-radical substitution, which we’ve seen is a pretty poorly selective reaction, (link), and 2) combustion, which kind of defeats the whole point of synthesis in the first place.
You know what is exciting, though? Reactions of alkenes.
It would be really useful to get our alkyne to accept only one equivalent of H2. Then we’d have an alkene, and we’d unlock the many useful reactions of alkenes we’ve learned previously. [See Article - Reaction Map of Alkenes to give you a fuller perspective] This would allow us to build in all kinds of functional groups - alcohols, alkyl halides, epoxides, and many more - that just aren’t readily available from boring ol’ alkanes. [Note 1]
As it turns out, alkynes are more reactive towards hydrogenation than alkenes. It is possible to get an alkyne to accept just one equivalent of H2, if one is very careful with doling out a single molar equivalent using a gas buret or similar.
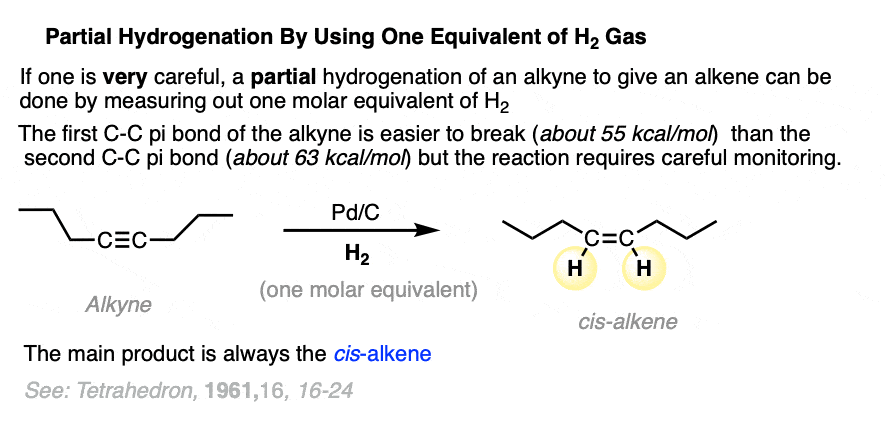
Note that while I say it is possible, it’s not actually all that convenient, since you need to set up an additional piece of equipment, and furthermore it can be tricky to accurately measure out one molar equivalent of H2 on small scale.
A simpler alternative for partial hydrogenation is just to make the catalyst worse.
Wait, what?
Yes, make it worse. Since alkynes are already more reactive than alkenes, just poison the catalyst enough so that is unable to carry out the hydrogenation of alkenes. Problem solved!
The most commonly used “poisoned” catalyst is Lindlar’s catalyst, which is a mixture of palladium, calcium carbonate, and traces of lead (Pb). Quinoline, an aromatic amine, is sometimes added to help with selectivity. . [Described in more detail here Note 2].
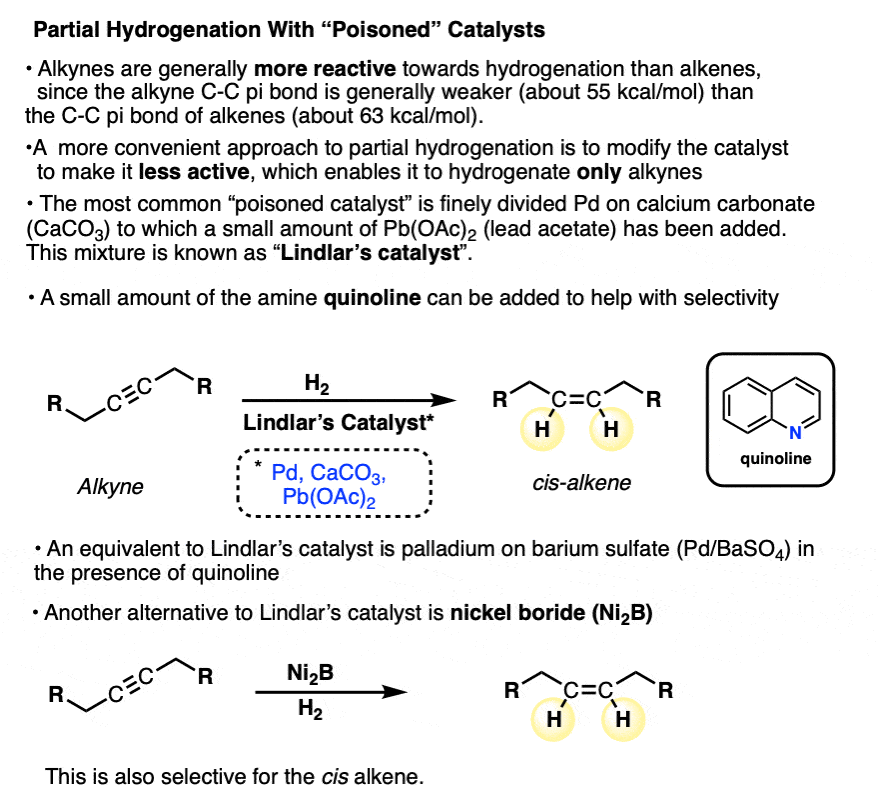
The “poisons” here are a lead (Pb) and amine [quinoline]. It is thought that lead (Pb) acts to reduce the bonding of H2 to the metal, while the amine (quinoline) helps to avoid unwanted byproducts. (See reference here)
Another reagent that sometimes sees use is nickel boride (“Ni2B”), which for our purposes can be considered to be equivalent to Lindlar’s catalyst. It is used in the presence of hydrogen gas. [Note 3]
2. Lindlar’s Catalyst: Examples
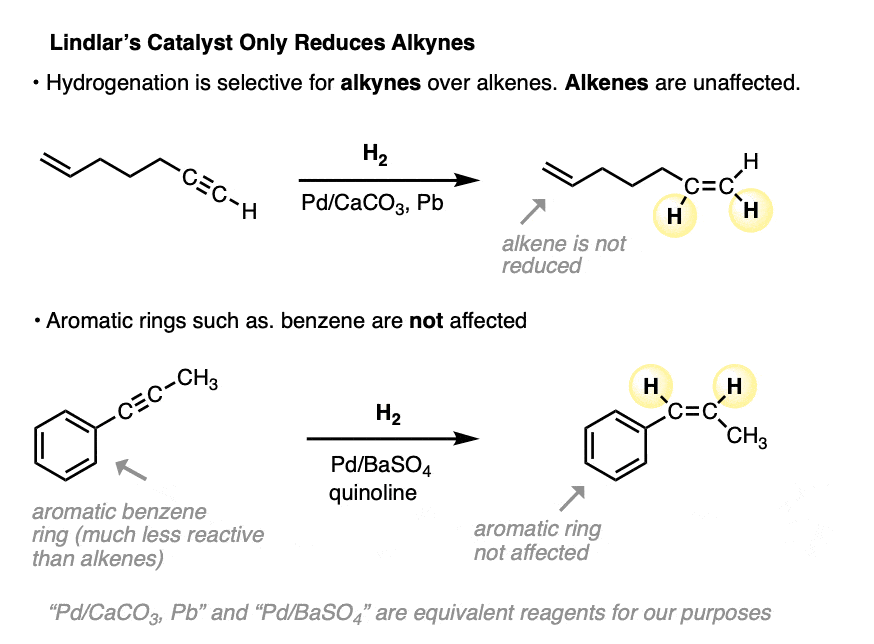
One advantage of Lindlar’s catalyst is that it is selective for the reduction of alkynes. Alkenes are not affected. The stereochemistry of the alkene produced is always cis.
Furthermore, aromatic rings such as benzene are also unaffected (the unusually low reactivity of benzene is covered in a subsequent chapter - see Introduction to Aromaticity).
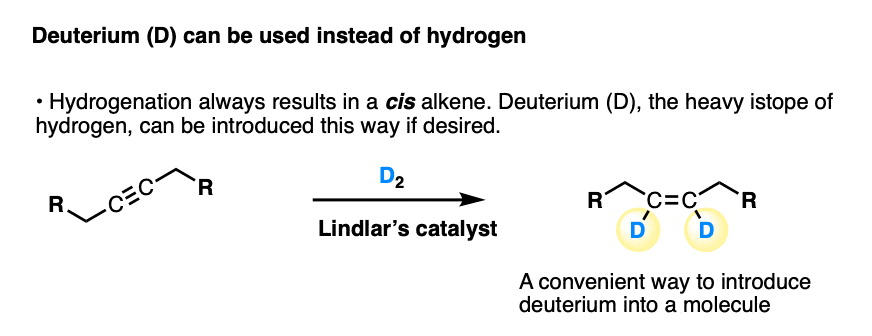
Deuterium, the heavy isotope of hydrogen, can be used in place of hydrogen gas.
3. Applications of Lindlar’s Catalyst
As mentioned above, a key feature of partial hydrogenation of alkynes is that it unlocks all the reactions of alkenes.
For example, alkynes don’t undergo epoxidation with m-CPBA or dihydroxylation with OsO4. But alkenes do!
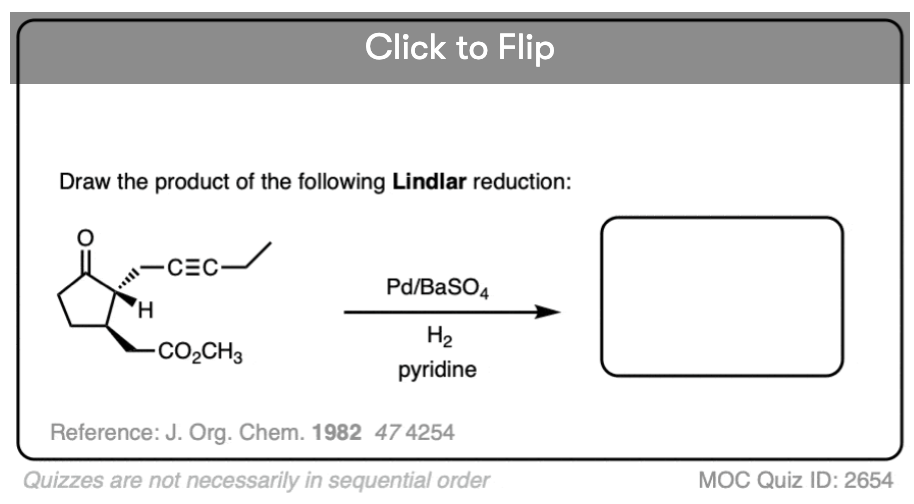
See if you can draw the product that is formed when an alkyne is partially hydrogenated with Lindlar’s catalyst and then treated with OsO4.
In these types of quiz questions, it’s very important to pay close attention to the stereoselective nature of the Lindlar reduction.
Lindlar’s catalyst is notable in that it makes cis-alkenes from alkynes. Another reagent, sodium in ammonia (Na/NH3) can reduce alkynes to trans-alkenes. (See article - Reduction of Alkynes to trans-Alkenes With Na/NH3).
Keeping stereochemistry straight is extremely important both in the forward (i.e. predicting the products) and reverse directions (e.g. synthesis problems, see below).
Try this example with halogenation.
Here is an example incorporating the addition of HBr :
Yes - even though we’re in the chapter on alkynes, Markovnikov’s rule still applies :-) .
Finally, an epoxidation example:
4. Synthesis Questions With Lindlar’s Catalyst
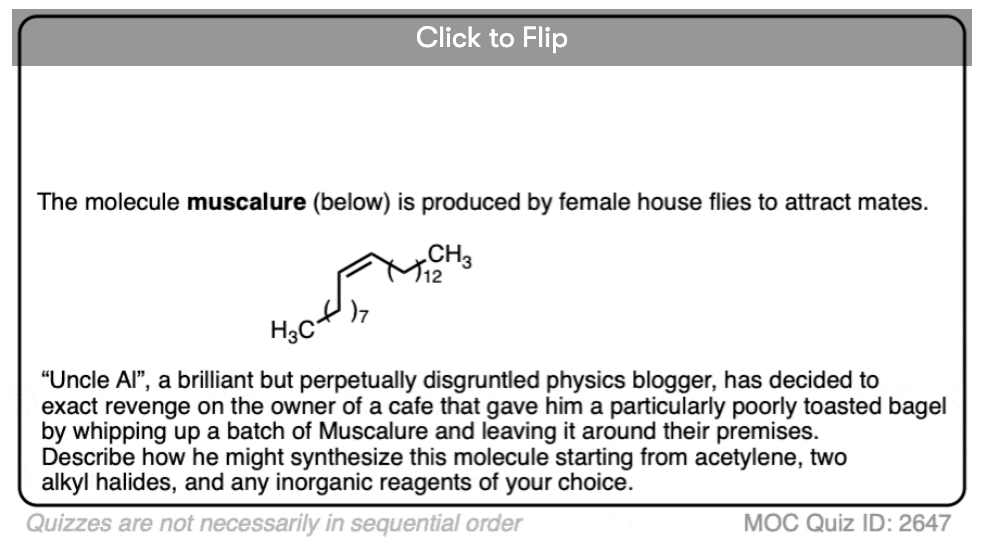
Now that we have a reaction for building up the carbon chain (acetylide alkylation) and a reaction for converting alkynes to alkenes (partial hydrogenation) a whole world is opening up before us.
We can finally think about how we would carry out the synthesis of larger molecules from smaller molecules through a series of well-planned reactions.
It’s like putting together Lego bricks, but instead of pushing Lego nubs into the holes of another brick, we’ll be adding nucleophiles to electrophiles.
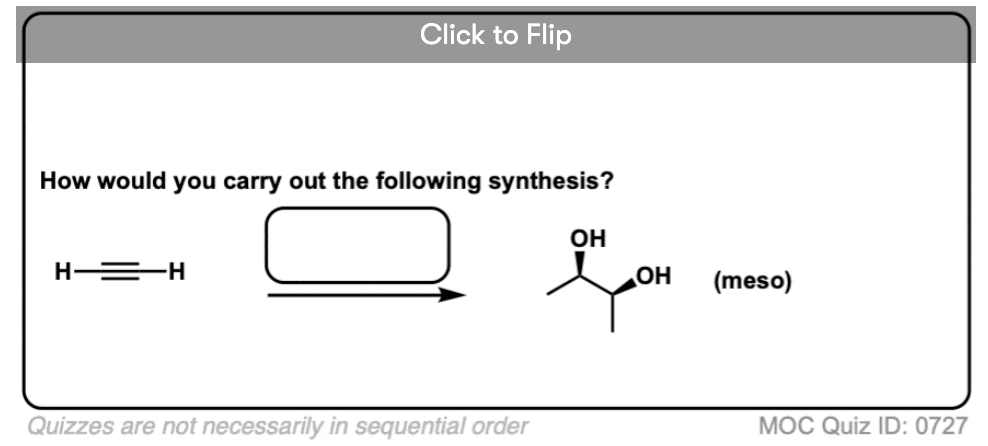
Here’s a typical example. How would you synthesize this diol from the alkyne (below left) using any inorganic reagents and carbon fragments with two carbons or less?
Working on these types of problems takes some practice. I’ll have more to say about it elsewhere, but when working on these problems, I advise breaking it down to the following exercises:
- Examine what’s new - what specific bonds have formed (and broken) in going from the starting material to the product? This is your to-do list.
- What reactions do you know that form and break those bonds?
- In what order do we need to do these reactions?
Once you’ve answered these questions, you can start thinking about how you would carry out the sequence in a forward direction.
One key factor not to be forgotten is the importance of stereochemistry.
When planning out these reactions, it’s not enough to know that, say, Lindlar’s catalyst does the partial reduction of an alkyne to an alkene. It’s crucial to also include that the reduction occurs in a cis fashion. If you leave out that detail, you won’t get full credit for your answers.
Here’s another example of a synthesis problem involving epoxidation (m-CPBA).
Here’s another example of a synthesis question that involves chlorination (remember that this is selective for the anti-addition product.
In this example, we had a vicinal dihalide that was drawn in a conformation where both C-Cl bonds were wedges. If we worked backwards from this without accounting for the stereochemistry, we’d end up with the wrong double-bond geometry.
5. Summary
Lindlar’s catalyst is used for the partial reduction of alkynes to cis-alkenes. Be on the lookout for synthesis questions involving Lindlar’s catalyst and pay close attention to stereochemistry.
In the next article we will cover a reaction for the partial reduction of alkynes to give trans alkenes, Na/NH3 (sodium in ammonia).
Notes
Note 1. One of the outstanding challenges in modern organic chemistry - a “holy grail“, if you will - is actually how to take a relatively unreactive functional group like an alkane C-H bond and transform it to a more reactive functional group such as C-OH or C-Cl with high selectivity. This extremely active area of research is known as “C-H activation“.
Note 2. Technically, Lindlar’s catalyst is Pd on calcium carbonate that has been treated with a trace amount of lead salt (the procedure is here). It has been found that addition of small amounts of quinoline also helps with selectivity (so do certain thiols).
Note 3. Nickel boride is made through the reduction of Ni(II) acetate with sodium borohydride (NaBH4) in methanol. It is a hydrogenation catalyst. A representative procedure can be found here.
Quiz Yourself!
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
(Advanced) References and Further Reading
- Catalytic Semihydrogenation of the Triple Bond Elliot N. MARVELL*, Thomas LI Synthesis 1973, 457-468 DOI: 10.1055/s-1973-22234 Fairly comprehensive review, still relevant today, on various methods for partial hydrogenation of alkynes.
- Ein neuer Katalysator für selektive Hydrierungen H. Lindlar Helv. Chim. Acta 1952 35 (2), 446 DOI: 10.1002/hlca.19520350205 The original paper by Lindlar describing the development of a new catalyst for the selective hydrogenation of alkynes to Z-alkenes during Vitamin A synthesis.
- PALLADIUM CATALYST FOR PARTIAL REDUCTION OF ACETYLENES H. Lindlar, R. Dubuis Org. Synth. 1966, 46, 89 DOI: 10.15227/orgsyn.046.0089 This procedure by Lindlar also gives a detailed preparation of the catalyst.
- A density functional theory study of the ‘mythic’ Lindlar hydrogenation catalyst Garcı´a-Mota, J. Gomez-Dı´az, G. Novell-Leruth, C. Vargas-Fuentes, L. Bellarosa, B. Bridier, J. Pe´rez-Ramı´rez, N. Lo´pez Theor. Chem. Acc. 2011, 128, 663 DOI: 10.1021/s00214-010-0800-0 This is a computational investigation using DFT (density functional theory) which studies how the various components in the Lindlar catalyst (Pd, Pb, quinoline) pack together and how that contributes to hydrogenation selectivity.
- (Z)-4-(TRIMETHYLSILYL)-3-BUTEN-1-OL L.E. Overman, M. J. Brown, S. F. McCann Org. Synth. 1990, 68, 182 DOI: 10.15227/orgsyn.068.0182 The second reaction in this 2-step synthesis is a Lindlar hydrogenation to give the Z-alkene.
- SYNTHETICALLY USEFUL REACTIONS WITH NICKEL BORIDE. A REVIEW Jitender M. Khurana, Amita Gogia. Organic Preparations and Procedures International The New Journal for Organic Synthesis DOI: 1080/00304949709355171 This is a review on the application of nickel boride in organic synthesis, which can be used in similar applications to Lindlar’s catalyst.
- Selective catalytic hydrogenation of acetylenes, N.A. Dobson, G. Eglinton, M. Krishnamurti, R.A. Raphael, R.G. Willis, Tetrahedron, 1961,16, 16-24 DOI: 10.1016/0040-4020(61)80050-1 In this article the authors establish that terminal alkynes are more reactive towards hydrogenation than internal alkynes, and internal alkynes are more reactive towards hydrogenation than alkenes. They also demonstrate that alkynes can be selectively hydrogenated by limiting the number of equivalents of hydrogen to one equivalent.
- Catalytic hydrogenation. VI. Reaction of sodium borohydride with nickel salts in ethanol solution. P-2 Nickel, a highly convenient, new, selective hydrogenation catalyst with great sensitivity to substrate structure Charles Allan Brown and Vijay K. Ahuja The Journal of Organic Chemistry 1973 38 (12), 2226-2230 DOI: 10.1021/jo00952a024 This work introduces P-2 nickel boride for the reduction of alkynes to cis-alkenes.
Link nội dung: https://hnou.edu.vn/c2h2-h2-pd-pbco3-a24551.html